 These extra neutrons are necessary for stability of the heavier nuclei. It can be observed from the chart that there are more neutrons than protons in nuclides with Z greater than about 20 (Calcium). This chart shows a plot of the known nuclides as a function of their atomic and neutron numbers. Also to help understand this concept there is a chart of the nuclides, known as a Segre chart. To determine the stability of an isotope you can use the ratio neutron/proton (N/Z). To identify the stability of an isotope it is needed to find the ratio of neutrons to protons. Nuclear stability is a concept that helps to identify the stability of an isotope. Neutron and Atomic Numbers and Nuclear Stability On the other hand, nuclei with an odd number of protons and neutrons are mostly unstable. Heavy nuclei with an even number of protons and an even number of neutrons are (due to Pauli exclusion principle) very stable thanks to the occurrence of ‘paired spin’. For example, actinides with odd neutron number are usually fissile (fissionable with slow neutrons) while actinides with even neutron number are usually not fissile (but are fissionable with fast neutrons). 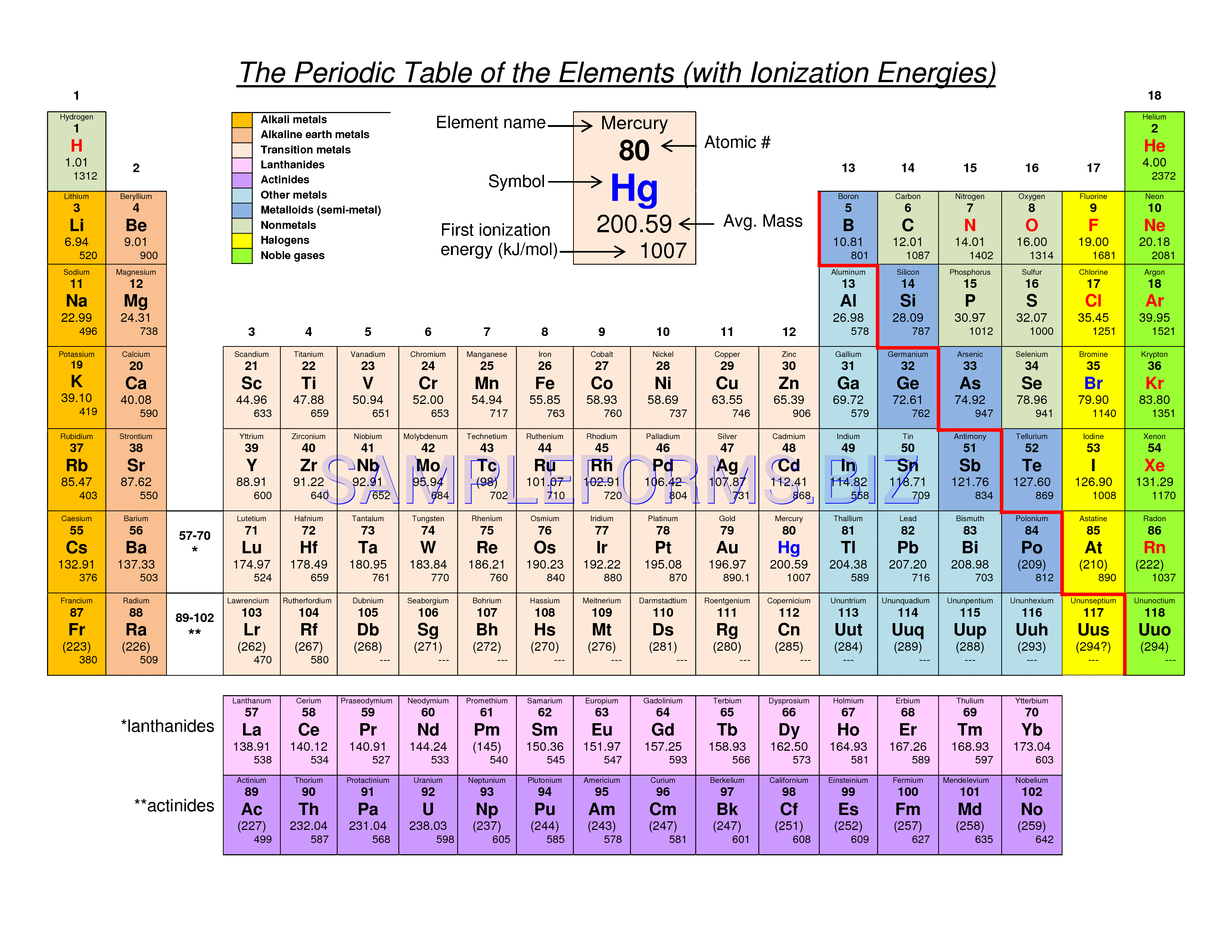
It must be noted, especially nuclear cross-sections may vary by many orders from nuclide with the neutron number N to nuclide with the neutron number N+1. Properties of atomic nuclei (atomic mass, nuclear cross-sections) are determined by the number of protons and number of neutrons (neutron number). Neutron and Mass Numbers and Nuclear Properties For example, the neutron number of uranium-238 is 238-92=146. We can determine the neutron number of certain isotope. Therefore, we cannot determine the neutron number of uranium, for example. Each nuclide is denoted by chemical symbol of the element (this specifies Z) with tha atomic mass number as supescript. The various species of atoms whose nuclei contain particular numbers of protons and neutrons are called nuclides. Nuclides that have the same neutron number but a different proton number are called isotones. Neutron number is rarely written explicitly in nuclide symbol notation, but appears as a subscript to the right of the element symbol. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. It has a melting point of 98°c and a boiling point of 883°c.Mass numbers of typical isotopes of Sodium are 23. It is a metallic solid at room temperature that is easily cut by a knife. It is located in Group 1, the Alkali metals. 
Sodium was discovered and isolated by Humphry Davy in 1807 who made it by separating sodium hydroxide using electrolysis. Sodium chloride is a common salt that is used widely in food preparation and also chemical reactions. Sodium Hydroxide is a widely used reagent in many chemical reactions as an alkali. However, it is the compounds of Sodium that have the most uses. Sodium metal itself has some uses in nuclear reactors and as a reagent in some chemical reactions due to its reactivity. Sodium is present in most living organisms and plays a large part in nerve transmissions in organisms. Much like Lithium, it is stored under oil to prevent oxidation by moisture and oxygen. It is an Alkali metal which will readily react with water to produce sodium hydroxide and also easily tarnishes in oxygen. Sodium is a highly reactive metal in Group 1 of the periodic table. It is an Alkali Metal and is located in Group 1 of the periodic table. 
Sodium (Na) is a silver-white coloured metal that has the atomic number 11 in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
